Recently, the research team led by Associate Professor Quan Jing from the College of Biological Science and Medical Engineering of Donghua University, in collaboration with the team led by Director Sun Peng from Tong Ren Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, published a research paper titled "CaO₂-based AuPt Bimetallic Nanozymes with Cascade Catalysis for Synergistic Chemodynamic/Photothermal/Immunotherapy" in the Chemical Engineering Journal, offering a novel therapeutic strategy for tumor treatment.

Nanozyme-based chemodynamic therapy (CDT) has become a research hotspot in the field of tumor treatment in recent years, owing to its advantages such as high safety, no need for exogenous oxygen supply, and the ability to modulate the tumor microenvironment. This therapeutic approach relies on Fenton or Fenton-like reactions to convert endogenous H₂O₂ within the tumor into highly toxic ROS to kill tumor cells. However, its clinical translation faces two major bottlenecks: insufficient catalytic activity of nanozymes and extremely low H₂O₂ concentration (only 50–100 × 10⁻⁶ M) in the tumor microenvironment, which makes it difficult to generate sufficient ROS. Moreover, a single treatment modality cannot completely eradicate tumors or prevent recurrence and metastasis, severely limiting clinical application. Therefore, the construction of highly efficient self-supplying H₂O₂ nanozyme systems and the integration of multimodal synergistic therapy have become key research directions in tumor nanomedicine. To address these challenges, the research team has carried out collaborative research through the integration of medicine and engineering, developing a calcium peroxide (CaO₂)-based AuPt bimetallic self-cascade nanozyme system, designated CaO₂/AuPt@BSA (CAPB), which enables triple synergistic antitumor therapy combining chemodynamic, photothermal, and immunotherapeutic effects. The corresponding authors of the paper are Quan Jing and Sun Peng, and the co-first authors include graduate students Liu Wentao and Zhu Lili from the College of Biological Science and Medical Engineering.
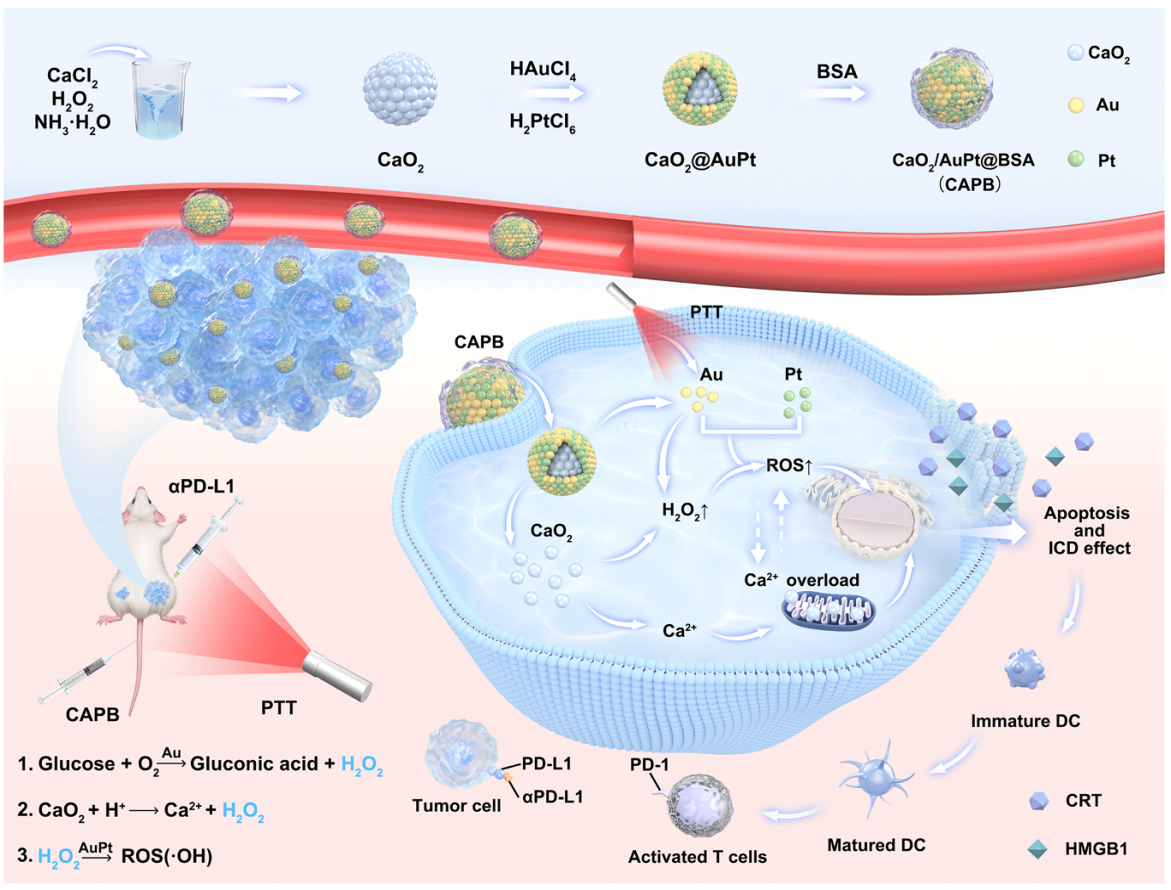
Scheme 1 (a) Schematic illustration of the preparation of CAPB nanozymes;
(b) Mechanism of action for tumor therapy
This CAPB nanozyme system is designed based on the acidic and metabolic characteristics of the tumor microenvironment. It employs a calcium chloride method to construct a biocompatible CaO₂ nanosphere core, integrates Au and Pt bimetallic nanozymes, and achieves excellent tumor targeting and physical stability through BSA surface encapsulation, with an average particle size of approximately 200 nm and stability maintained for 7 days. The system precisely targets tumors via efficient self-cascade catalytic reactions: upon entering the acidic tumor microenvironment, the Au nanozyme depletes glucose to induce "starvation therapy" and generates H₂O₂; subsequently, the Pt nanozyme catalyzes the conversion of H₂O₂ into highly toxic hydroxyl radicals (•OH) to enhance chemodynamic therapy (CDT). Meanwhile, the Ca²⁺ released from CaO₂ decomposition can induce severe mitochondrial dysfunction.
In terms of energy conversion and catalytic performance, the Au nanozyme also endows this system with excellent photothermal conversion efficiency (36.3%). Under 808 nm laser irradiation, it induces a significant temperature rise (approximately 38 °C for a 400 μg/mL solution), enabling not only photothermal ablation (PTT) of tumors but also marked acceleration of the cascade catalytic reaction kinetics (Km = 39.178 ± 7.656 mM, Vmax = 2.249×10⁻⁶ ± 1.923×10⁻⁷ M min⁻¹). In vitro experiments confirm that the system specifically recognizes and kills 4T1 tumor cells, achieving an apoptosis rate of 58.8% under photothermal enhancement, while exhibiting low cytotoxicity against normal cells.
Regarding in vivo antitumor efficacy and immune activation, the CAPB nanozyme demonstrates potent synergistic therapeutic capability. Animal studies show that the nanozyme accumulates at the tumor site 8 hours post-administration. When combined with αPD-L1 antibody therapy, it achieves a primary tumor inhibition rate of 82.7%, which is 1.67 times that of the immunotherapy-alone group, and a distant tumor inhibition rate of 79.18%. This remarkable efficacy is attributed to immunogenic cell death (ICD) synergistically induced by CDT and PTT, which effectively promotes the release of damage-associated molecular patterns (DAMPs). Consequently, the proportions of mature dendritic cells (DCs) and CD8⁺ T cells in vivo are increased by 2.60-fold and 1.73-fold, respectively, compared with the control group, accompanied by upregulation of key immune factors such as TNF-α and IL-12. This triple synergistic strategy integrating catalytic, photothermal, and immunotherapeutic actions significantly suppresses tumor growth and metastasis while demonstrating favorable biosafety, providing strong scientific evidence and application prospects for multimodal synergistic tumor therapy.
This research was supported by several funding sources, including the "Shanghai Tongren Hospital - Donghua University Medical-Engineering Cross-Disciplinary Project", demonstrating the significant role and value of in-depth hospital-university collaboration in driving medical technology innovation and supporting high-quality regional development.
Original link to the article:https://doi.org/10.1016/j.cej.2026.17256
