Recently, Professor Jinli Qiao’s research team from the College of Environmental Science and Engineering has made significant progress in the field of proton exchange membrane fuel cells. The related findings were published in the internationally renowned journal in electrochemistry, Applied Catalysis B: Environmental and Energy, under the title “Synergistic Pt Single-Atom and Pt Nanoparticle Catalysts Anchored on N-Doped Mesoporous Carbon Architecture Boosting High-Performance Oxygen Reduction in PEMFCs.”

In this study, a zinc-assisted strategy was successfully employed to fabricate a platinum(Pt)-based heterostructured electrocatalyst, in which atomically dispersed platinum single atoms (PtSAs) and platinum nanoparticles (PtNPs) coexist within a nitrogen-doped mesoporous carbon matrix. This dual-active-site configuration maximizes atomic utilization, thereby bridging the performance gap between single-atom catalysts and conventional nanoparticle-based catalysts. The hybrid architecture exhibits exceptional oxygen reduction reaction activity, long-term durability, and high selectivity, with performance metrics far exceeding those of commercial Pt/C catalysts.
Proton exchange membrane fuel cells (PEMFCs) are widely regarded as a next-generation clean energy technology owing to their high energy efficiency, fast start-up capability, and near-zero greenhouse gas emissions. These characteristics make them key enablers in the global transition toward sustainable energy systems. Platinum-based nanomaterials currently remain the benchmark electrocatalysts for the oxygen reduction reaction(ORR) in proton exchange membrane fuel cells(PEMFCs). Among them, commercial Pt/C catalysts are extensively utilized due to their high catalytic activity and well-established technological framework. However, achieving high activity, high durability, and high metal utilization simultaneously remains a critical challenge in this field.
The heterostructured electrocatalyst (PtSA-NP/MPC950) developed in this work achieves a half-wave potential of 0.90 V vs. RHE, a mass activity of 1.26 A mgPt⁻¹ (6.3 times that of Pt/C), and a 42% improvement in durability after 50,000 cycles in acidic media. The hierarchical porous carbon support enables efficient mass transport, while the synergistic interaction between platinum single atoms and nanoparticles optimizes intermediate binding—thereby accelerating ORR kinetics and suppressing catalyst degradation. Complementary density functional theory calculations further reveal that the synergy between single atoms and nanoparticles lowers the energy barrier of the rate-determining step and inhibits H₂O₂ formation, establishing a direct structure–property relationship. This work provides a generalizable design paradigm for fabricating high-loading, durable, and efficient electrocatalysts, thereby offering valuable insights for advancing next-generation proton exchange membrane fuel cell technologies and other electrochemical energy conversion devices.
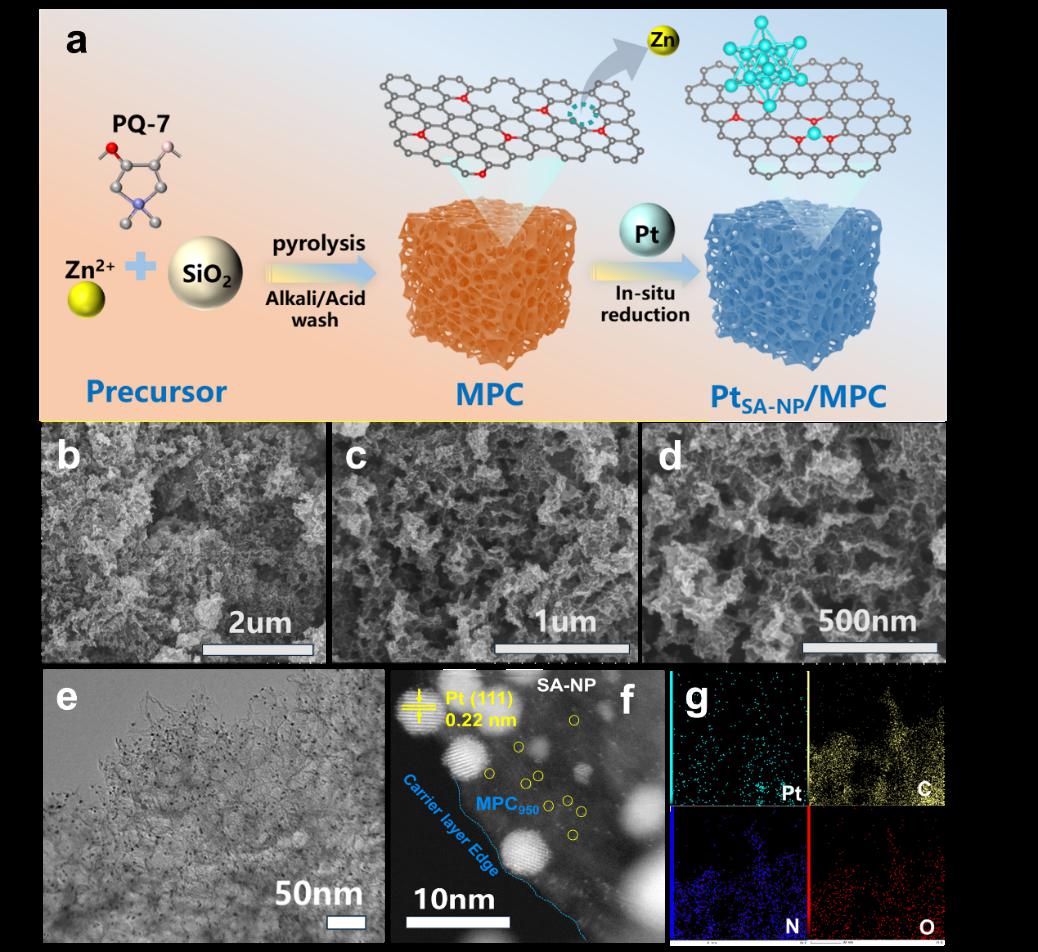
(synthesis and characterization of PtSA-NP/MPC950)
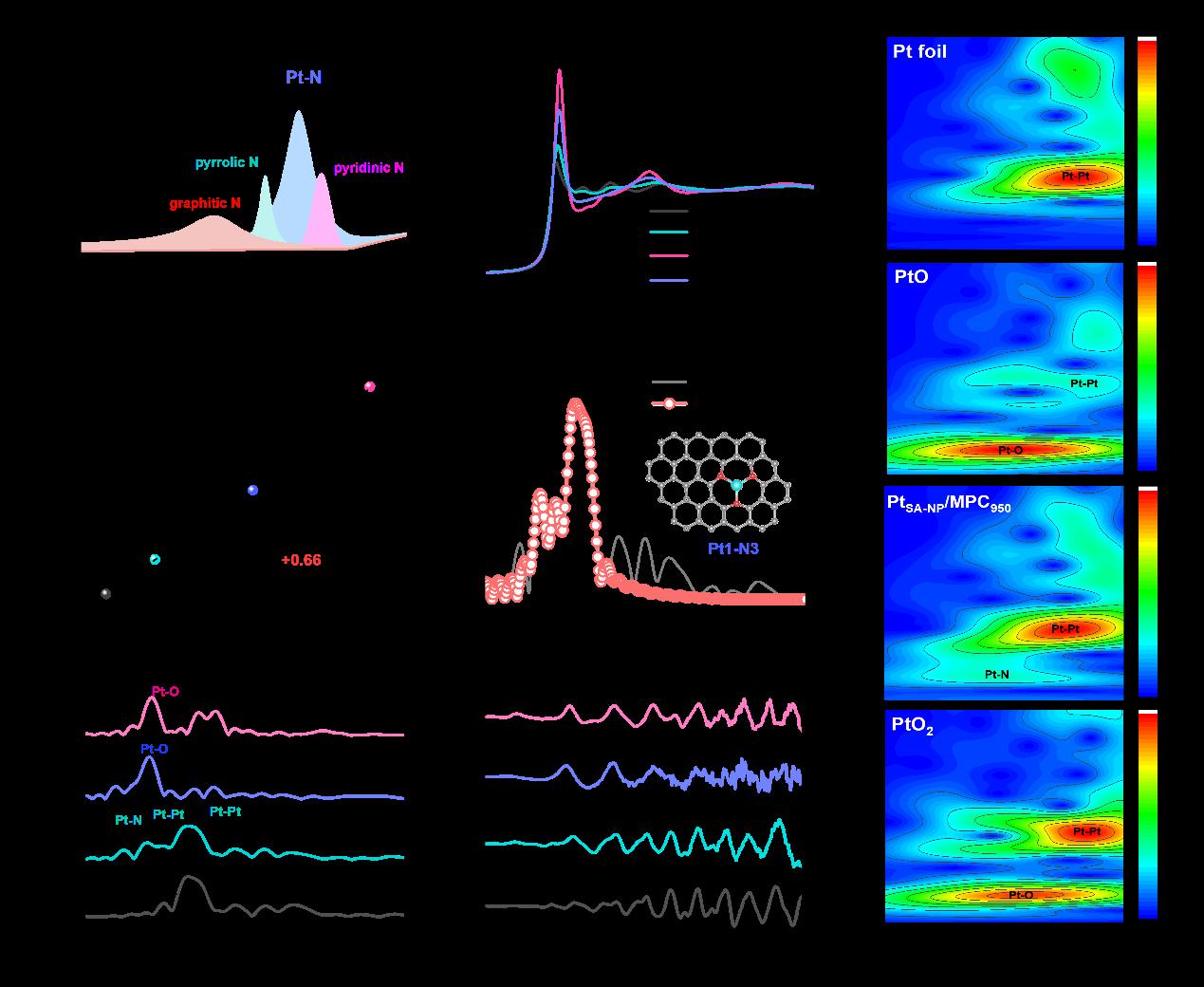
(Structural Characterization of Catalysts)
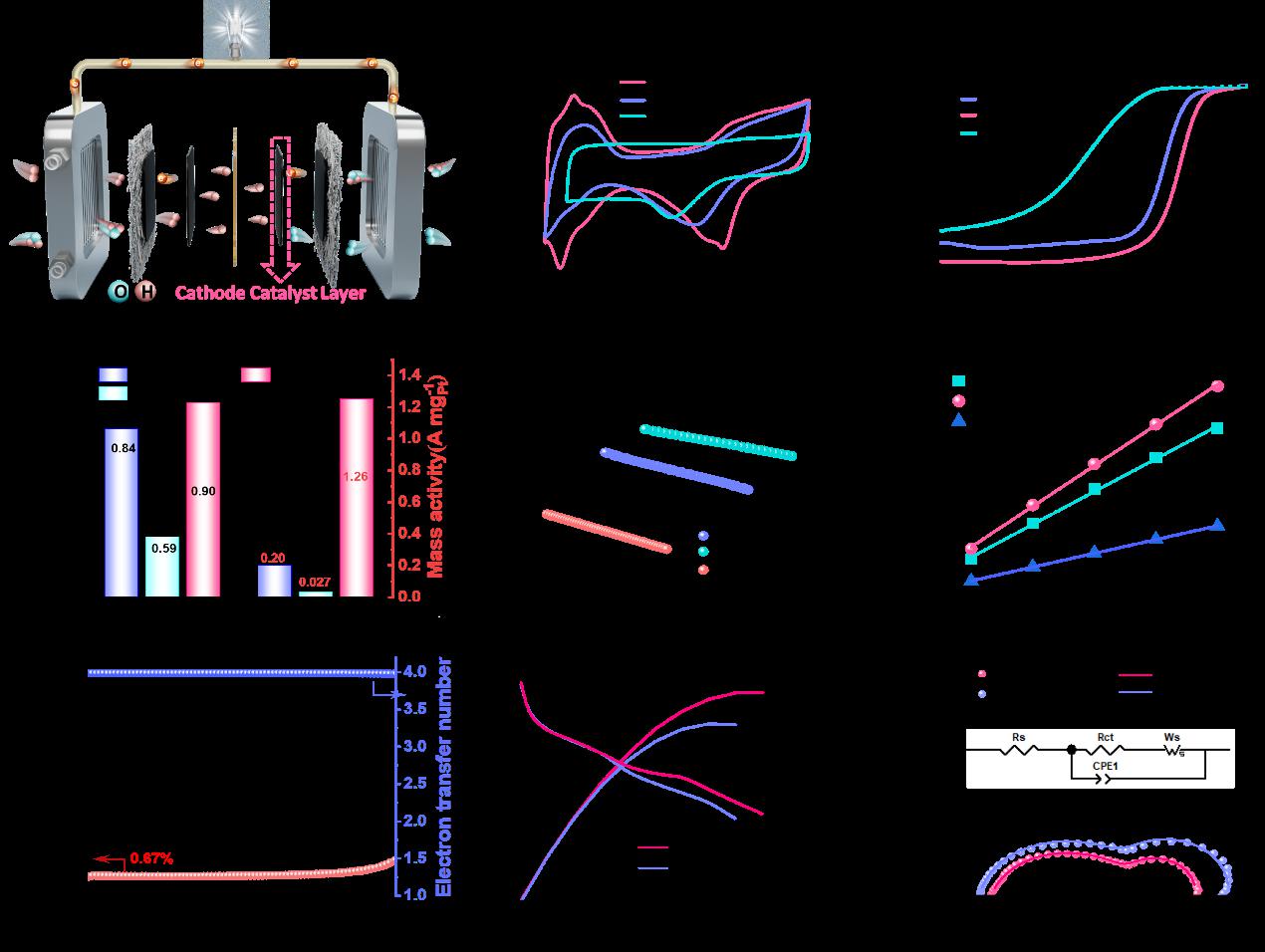
(Electrochemical Properties of MPC950, PtSA-NP/MPC950, and Commercial Pt/C)
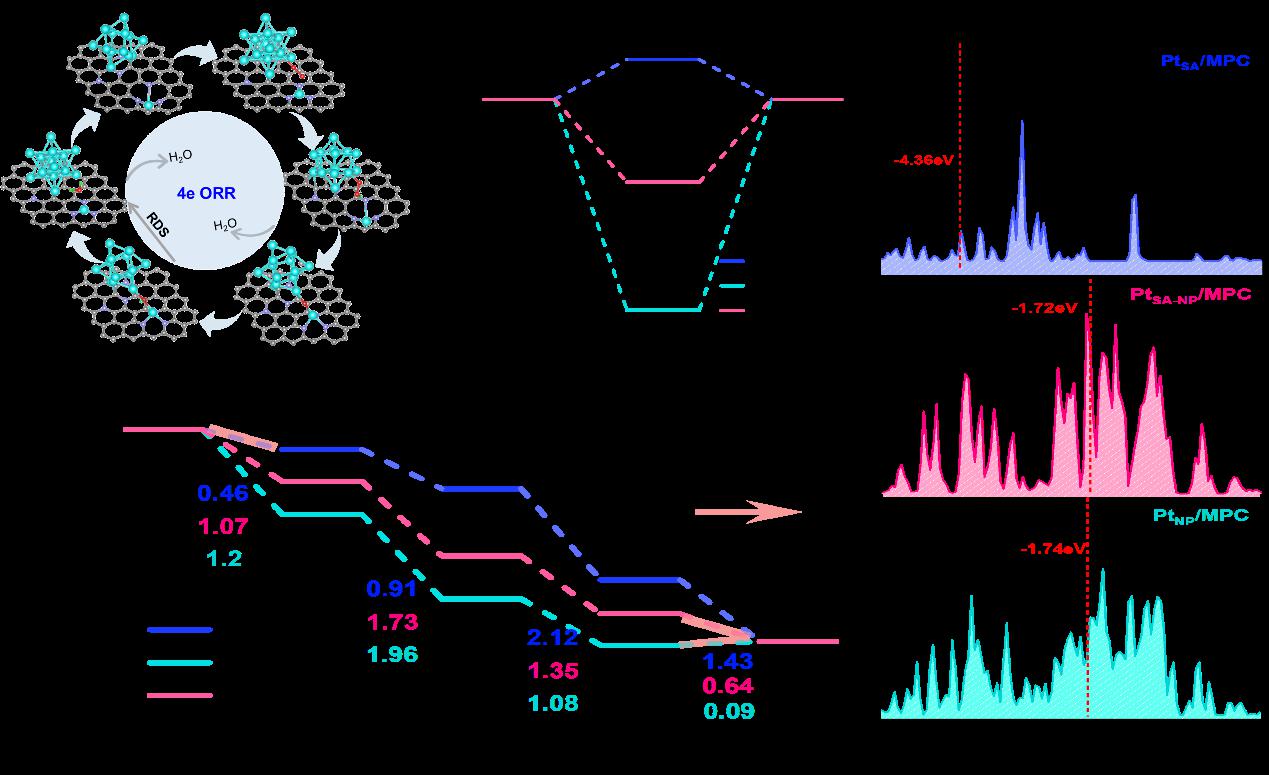
(DFT calculations of ORR for the PtSA/MPC, PtNP/MPC, and PtSA-NP/MPC models)
This work was supported by the Intergovernmental International Science and Technology Innovation Cooperation Project under the State Key Research and Development Program (2022YFE0138900) and the National Natural Science Foundation of China (21972017).
(Original link to this article:https://doi.org/10.1016/j.apcatb.2026.126408)
